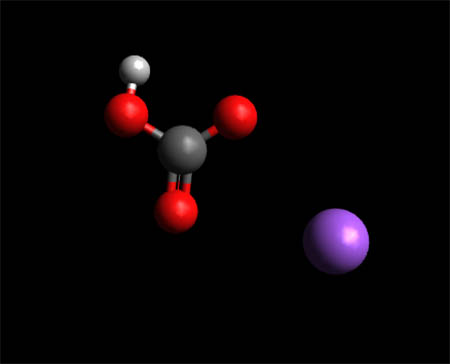
SOLVED: Determine the mass of baking soda (84 g/mol) needed to completely react with 355 mL of vinegar. Assume vinegar is 5% (m/m) acetic acid (60 g/mol) and the density of vinegar

Preparation and properties of sodium bicarbonate‐incorporated LDPE films with deodorizing function for kimchi packaging - Jeong - 2017 - Packaging Technology and Science - Wiley Online Library

Wuxi Beitang Chemical Light Raw Material Co., Ltd Sodium bicarbonate(baking soda)Wuxi Beitang Chemical Light Raw Material Co., Ltd

Tap Density Tester,Kit To Measure The Baking Soda Density - Buy Tap Density Tester,Kit To Measure The Baking Soda Density,Density Tester Product on Alibaba.com
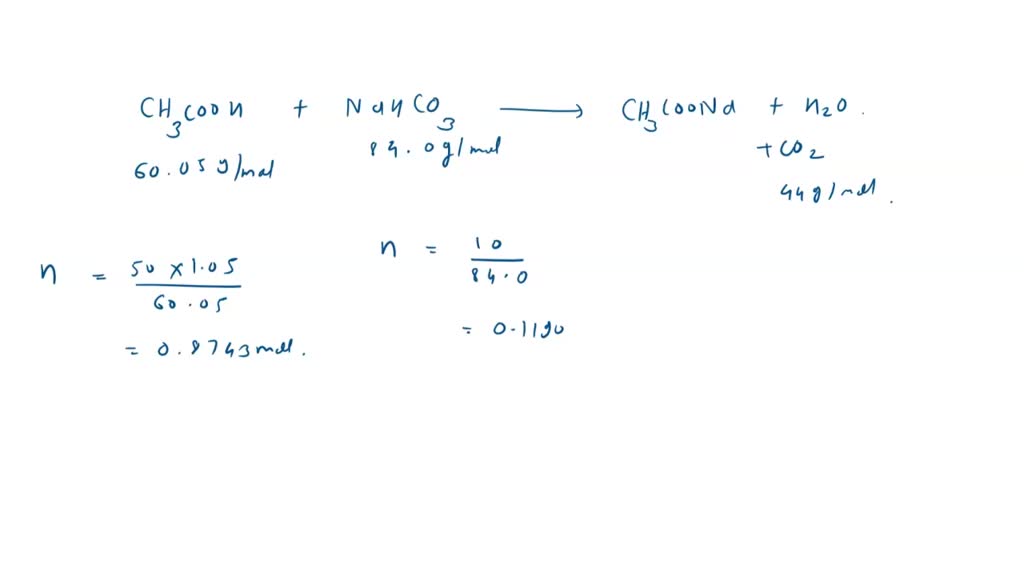
SOLVED: 10g of baking soda (Sodium Bicarbonate) is added to 50mL of vinegar (Density = 1.05g/mL). The resulting solution weighs 60.74g. How many liters of CO2 gas are released if it has

Water density science experiment || Water density changes when other substances dissolve in it - YouTube

SOLVED: 048 3.053 Datu: Mise baking soda (NaHCOz) used Mastt glove. baking soda vinegar REFORE MIXING Mass elove, baking soda vinegar AFTER MIXING Total volume of COz gas collected Nolumc afwatot bcakarl